Eager for hands-on learning with Lumenis’ award-winning, innovative technology? Join us at Brilliance and industry meetings to be part of the conversation.
Lumenis OptiLight Wins 2022 MedTech Breakthrough Award: Best New Technology Solution for Ophthalmology
For the third straight year Lumenis wins an award recognizing outstanding medical companies and technologies.
triLift
The triLift enables dynamic muscle stimulation with 3 different applicators to tone healthy muscles, reduce wrinkles, and achieve a face life like effect.
Hand in Hand: A Thriving Dry Eye Practice and Boutique Optical
Dr. Mei Fleming ensures her patients “See better than 20/20 in style” and get comprehensive dry eye disease management, including OptiLight from Lumenis.
Lumenis and Baring Private Equity Asia to Accelerate Investment in Aesthetics and Vision Verticals Following the Strategic Sale of Lumenis Surgical for $1.07B
Lumenis will refocus and accelerate planned investments to support the development of its fast-growing Aesthetic and Vision businesses, emphasizing R&D, global sales and marketing channels, and business development activities.
Diversify Your Practice for Long-term Success
Dr. Nate Lighthizer advises ODs to embrace management of eye diseases such as dry eye and glaucoma. Here are his tips for building a diverse, healthy optometry practice.
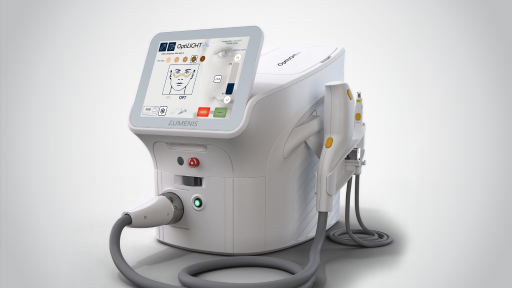
OptiLIGHT
Introducing OptiLIGHT – a bright solution for dry eyes. OptiLIGHT elevates dry eye management with Lumenis’ patented Optimal Pulse Technology (OPT™) and user-centered design. Get the safe, precise, elegant procedure you want and the comfortable, effective therapy your patients need to manage dry eye disease with OptiLIGHT.
Lumenis Receives FDA Approval for Its IPL Device to Manage Dry Eye Disease and Launches OptiLight™
Lumenis Ltd., the world’s largest energy-based medical device company for ophthalmic, aesthetic and surgical applications and the inventor of intense pulsed light (IPL) technology, today announced that the FDA has granted De Novo authorization for Lumenis’ newest IPL device for improving signs of dry eye disease due to meibomian gland dysfunction (MGD).
Baring Private Equity Asia Announces Agreement to Sell Lumenis’ Surgical Business to Boston Scientific for $1.07 bln
Baring Private Equity Asia (BPEA) and Lumenis announced today that they have entered into a definitive agreement to sell the Lumenis Surgical Business to Boston Scientific (NYSE: BSX) in a transaction valued at USD1.07 billion, subject to closing adjustments.
Stellar M22 Optimized Design Optimized Success
Want to learn how the Stellar M22 can drive your practice forward? With four technologies in one platform, Stellar M22 can treat over 30 skin conditions and hair removal.